RESOF SOFOSBUVIR TABLETS
RESOF SOFOSBUVIR TABLETS Specification
- Expiration Date
- 24 Months from Manufacturing
- Grade
- Pharmaceutical Grade
- Medicine Type
- Allopathic
- Indication
- Chronic Hepatitis C Virus Infection
- Dosage
- 400 mg
- Appearance
- Film Coated Tablet
- Storage
- Cool, Dry and Dark Place
- Salt Composition
- Sofosbuvir 400mg
- CAS No
- 1190307-88-0
- Usage
- Used in the treatment of chronic hepatitis C infection
- Origin of Medicine
- India
- Assay
- NLT 98% (By HPLC)
- Dosage Form
- Tablets
- Molecular Formula
- C22H29FN3O9P
- Pacakaging (Quantity Per Box)
- 28 Tablets
- Formulation
- Film Coated Tablets
- Trade Name
- RESOF
- Prescription/Non prescription
- Prescription
- Administration Route
- Oral
- Packaging Type
- Box
- Color
- Yellow
- Minimum Order Quantity
- 1 box
RESOF SOFOSBUVIR TABLETS Trade Information
- Packaging Details
- PACK OF 28 TABLETS
- Main Domestic Market
- All India
About RESOF SOFOSBUVIR TABLETS
Resof 400 Mg Sofosbuvir
Resof - Sofosbuvir
Resof Contains Sofosbuvir 400 mg, Sofosbuvir Is Used for Treating Chronic Hepatitis C Virus (HCV) Infection
Specifications :-
- Brand Name :- Resof
- Content :- Sofosbuvir
- Company :- Dr. Reddys
- Strength :- 400mg
- Packing :- 1X28 tablets
- Type :- Tablet.
Note : - Resof Is A Prescription Drug And Should Be Used Under Proper Medical Guidance And Advice. Do Not Share The Medicine With Others, Since They May Be Suffering From A Problem That Is Not Effectively Treated By This Drug............
Precise Film-Coated Formulation
RESOF tablets feature a film-coated design to facilitate easier oral administration and improve patient compliance. The 400 mg dose is precisely measured and verified to meet pharmaceutical grade standards, ensuring reliable treatment for chronic hepatitis C infection. Each tablet maintains a high level of purity, with an assay value of at least 98%.
Optimal Packaging and Storage Conditions
The product is available in a secure box, with each box containing 28 tablets. RESOF should be stored in a cool, dry, and dark place to preserve its potency and stability throughout the 24-month shelf life. This packaging supports safe handling and distribution for medical professionals and providers.
Trusted Treatment for Hepatitis C
Sofosbuvir, the active ingredient in RESOF, is well recognized for its effectiveness in managing chronic Hepatitis C. The products oral route, precise salt composition, and Indian origin assure patients and healthcare providers of its quality and reliability as part of antiviral therapy regimens.
FAQs of RESOF SOFOSBUVIR TABLETS:
Q: How should RESOF Sofosbuvir Tablets be administered for hepatitis C treatment?
A: RESOF Sofosbuvir Tablets should be taken orally, as directed by a healthcare professional. The standard dosage is one 400 mg tablet daily, preferably at the same time each day.Q: What is the main benefit of using RESOF Sofosbuvir Tablets?
A: The primary benefit of RESOF Sofosbuvir Tablets is their efficacy in treating chronic Hepatitis C infection, helping to reduce viral levels and improve liver health over the prescribed course of treatment.Q: When should patients start using RESOF Sofosbuvir Tablets?
A: Patients should begin taking RESOF only after receiving a prescription from a qualified healthcare provider and a confirmed diagnosis of chronic Hepatitis C infection.Q: Where are RESOF Sofosbuvir Tablets manufactured and distributed?
A: RESOF Sofosbuvir Tablets are manufactured in India and are distributed, exported, supplied, traded, and wholesaled by reputable pharmaceutical distributors across various regions.Q: What is the recommended storage condition for RESOF Sofosbuvir Tablets?
A: For optimal effectiveness, RESOF should be stored in a cool, dry, and dark place. Proper storage helps maintain tablet quality and extends shelf life up to 24 months from the manufacturing date.Q: How can healthcare providers order RESOF Sofosbuvir Tablets?
A: Healthcare providers can place a minimum order of one box (containing 28 tablets) through authorized distributors or suppliers, ensuring the medication is sourced from certified channels.Q: What assurance of quality does RESOF offer regarding its formulation?
A: RESOF Sofosbuvir Tablets are formulated with pharmaceutical-grade quality, maintaining an assay of not less than 98% by HPLC and containing accurately measured Sofosbuvir 400mg per tablet.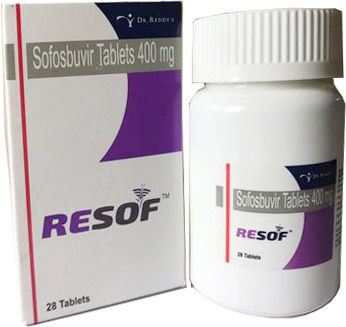

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in SOFOSBUVIR Category
HEPCINAT 400MG SOFOSBUVIR TABLETS
Medicine Type : Allopathic
Grade : Pharmaceutical Grade
Appearance : Filmcoated tablets
Dosage : 400 mg
Storage : Store below 30C
Origin of Medicine : India
Pharma Heptos RIBAVIRIN 200MG CAPSULES
Medicine Type : Allopathic
Grade : Pharmaceutical Grade
Appearance : White to offwhite powder (encapsulated)
Dosage : 200mg
Storage : Store below 30C, protect from moisture
Origin of Medicine : India
HEPCINAT 400 MG SOFOSBUVIR TABLETS
Medicine Type : Allopathic
Grade : Pharmaceutical Grade
Appearance : Filmcoated tablet
Dosage : 400 mg
Storage : Store below 30C, protect from light and moisture
Origin of Medicine : India
SOFOVIR SOFOSBUVIR 400 MG TABLETS
Medicine Type : Allopathic
Grade : Pharmaceutical Grade
Appearance : White to offwhite tablets
Dosage : 400 mg
Storage : Store below 30C, protected from moisture
Origin of Medicine : India
 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry