HEPCVIR L SOFOSBUVIR AND LEDIPASVIR TABLETS
HEPCVIR L SOFOSBUVIR AND LEDIPASVIR TABLETS Specification
- Dosage Form
- Tablet
- Pacakaging (Quantity Per Box)
- 28 Tablets
- Origin of Medicine
- India
- Indication
- Chronic Hepatitis C Virus (HCV) infection
- Expiration Date
- 24 to 36 months from manufacturing date
- Grade
- Pharmaceutical Grade
- Salt Composition
- Sofosbuvir and Ledipasvir
- Usage
- For the treatment of chronic hepatitis C infection
- Medicine Type
- Allopathic
- Assay
- Sofosbuvir 400 mg + Ledipasvir 90 mg
- Dosage
- One tablet daily or as directed by physician
- Storage
- Store below 30C, protected from moisture and light
- CAS No
- 1190307-88-0 (Sofosbuvir); 1256388-51-8 (Ledipasvir)
- Molecular Formula
- Sofosbuvir: C22H29FN3O9P; Ledipasvir: C49H54N8O6
- Appearance
- Film-coated tablets
- Shelf Life
- 2 to 3 years
- Product Name
- HEPCVIR L
- Prescription Status
- Prescription Only
- Therapeutic Class
- Antiviral agent
- Intended Patient Population
- Adults with genotype 1, 4, 5, or 6 HCV infection
- Marketed In
- India and export markets
- Manufactured By
- Cipla Ltd.
- Administration Route
- Oral
- Packing Type
- Bottle
- Colour
- Yellow to orange
- Drug Class
- NS5A inhibitor (ledipasvir) and nucleotide analog inhibitor (sofosbuvir)
HEPCVIR L SOFOSBUVIR AND LEDIPASVIR TABLETS Trade Information
- Packaging Details
- PACK OF 28 TABLETS
- Main Domestic Market
- All India
About HEPCVIR L SOFOSBUVIR AND LEDIPASVIR TABLETS
Hepcvir L Sofosbuvir & Ledipasvir
Specifications:
- Brand Name : Hepcvir L
- Content: Sofosbuvir and Ledipasvir
- Strength: Sofosbuvir 400 mg & Ledipasvir 90 mg
- Packing: 1X28 Tablets
- Type: Tablets
- Uses: Hepatitis C
- Manufactured By : Cipla
Note :-Hepcvir L Is A Prescription Drug And Should Be Used Under Proper Medical Guidance And Advice. Do Not Share The Medicine With Others, Since They May Be Suffering From A Problem That Is Not Effectively Treated By This Drug...............
Effective Antiviral Therapy for HCV
HEPCVIR L combines sofosbuvir and ledipasvir for a potent, targeted approach to treating chronic hepatitis C infection. This combination therapy blocks viral replication and significantly increases the chances of complete viral clearance, making it a reliable option for adult patients with specific HCV genotypes.
Convenient Dosage and Administration
HEPCVIR L comes in a bottle with 28 film-coated tablets. The recommended dosage is one tablet daily, taken orally, or as directed by your physician. The user-friendly, once-daily schedule helps support patient adherence throughout the treatment course, improving the likelihood of therapeutic success.
Quality and Safety Assured
Manufactured to pharmaceutical standards by Cipla Ltd., each batch of HEPCVIR L undergoes stringent quality control. With a shelf life of 2 to 3 years from manufacturing, the medicine retains its efficacy when stored properly. The formulation and packaging safeguard the stability of active ingredients.
FAQs of HEPCVIR L SOFOSBUVIR AND LEDIPASVIR TABLETS:
Q: How should HEPCVIR L be taken for optimal results?
A: HEPCVIR L should be taken exactly as prescribed by your healthcare provider, typically one tablet orally once a day. For best results, take the tablet at the same time each day and follow your physicians instructions closely throughout the treatment period.Q: What medical condition is HEPCVIR L used to treat?
A: HEPCVIR L is intended for the treatment of chronic hepatitis C virus (HCV) infection in adults, specifically for patients with genotypes 1, 4, 5, or 6.Q: When should I start seeing the effects of HEPCVIR L?
A: The timeline for improvement varies, but viral load reductions are often observed after several weeks of daily administration. Full course duration and monitoring will be determined by your doctor based on your individual response.Q: Where can I purchase HEPCVIR L?
A: HEPCVIR L is available by prescription only and can be obtained at licensed pharmacies or through authorized distributors, exporters, and suppliers in India and some export markets.Q: What is the process for storing HEPCVIR L safely?
A: Store HEPCVIR L in its original bottle below 30C, protected from moisture and light. Always keep the bottle tightly closed to maintain product quality and potency.Q: How is HEPCVIR L beneficial for hepatitis C patients?
A: By inhibiting key viral enzymes, HEPCVIR L disrupts the hepatitis C viruss ability to replicate, leading to significant improvements in liver health and a higher chance of achieving a sustained virologic response (cure).

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in SOFOSBUVIR Category
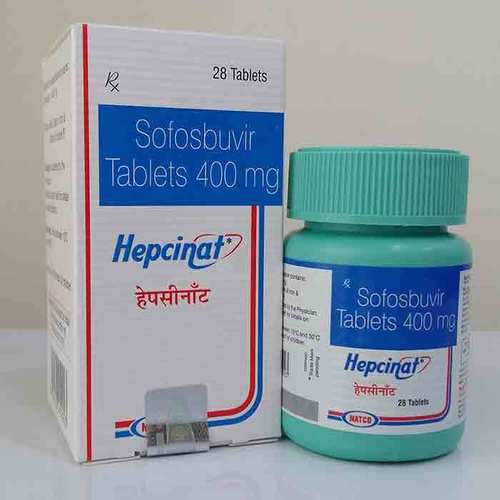
HEPCINAT 400MG SOFOSBUVIR TABLETS
Salt Composition : Sofosbuvir 400mg
Medicine Type : Allopathic
Usage : For the treatment of chronic Hepatitis C virus (HCV) infection
Grade : Pharmaceutical Grade
Expiration Date : 2436 months from manufacturing date
Indication : Chronic Hepatitis C
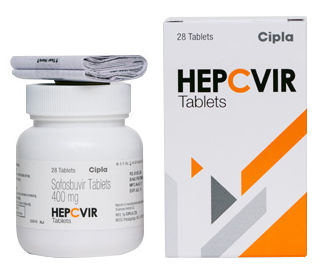
HEPCVIR SOFOSBUVIR TABLETS
Salt Composition : Sofosbuvir
Medicine Type : Allopathic
Usage : For the treatment of chronic hepatitis C infection
Grade : Pharmaceutical Grade
Expiration Date : 36 months from manufacturing date
Indication : Hepatitis C Virus (HCV) infection
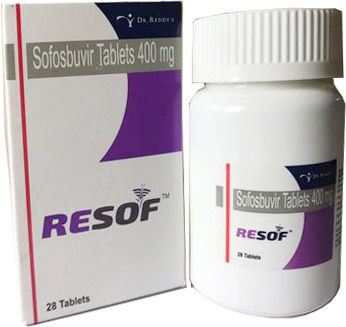
RESOF SOFOSBUVIR TABLETS
Salt Composition : Sofosbuvir 400mg
Medicine Type : Allopathic
Usage : Used in the treatment of chronic hepatitis C infection
Grade : Pharmaceutical Grade
Expiration Date : 24 Months from Manufacturing
Indication : Chronic Hepatitis C Virus Infection
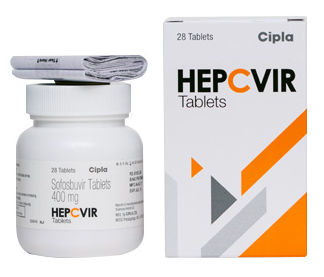
HEPCVIR SOFOSBUVIR 400MG TABLETS
Salt Composition : Sofosbuvir 400 mg
Medicine Type : Allopathic
Usage : Used for the treatment of chronic Hepatitis C infection
Grade : Medicine Grade
Expiration Date : 2436 months from manufacturing date
Indication : Chronic Hepatitis C Virus (HCV) infection
 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry