MYEZOM BORTEZOMIB 2 MG INJECTION
MYEZOM BORTEZOMIB 2 MG INJECTION Specification
- Salt Composition
- Bortezomib 2 mg
- Pacakaging (Quantity Per Box)
- 1 vial per box
- Indication
- For the treatment of Multiple Myeloma and Mantle Cell Lymphoma
- Dosage Form
- Lyophilized Powder for Injection
- Origin of Medicine
- Allopathic
- Drug Type
- Prescription Only
- Ingredients
- Bortezomib
- Physical Form
- Injection
- Function
- Recommended For
- Multiple Myeloma, Mantle Cell Lymphoma
- Dosage
- 2 mg per vial, as prescribed by physician
- Suitable For
- Adults
- Quantity
- 1 Vial
- Storage Instructions
- Store below 25C, protect from light
MYEZOM BORTEZOMIB 2 MG INJECTION Trade Information
- Packaging Details
- PACK OF 1 VIAL
About MYEZOM BORTEZOMIB 2 MG INJECTION
Myezom
This medication is a cancer chemotherapy drug used to treat certain types of tumors like multiple myeloma, mantle cell lymphoma. It blocks certain proteins which helps to slow the growth and spread of the cancer Available as: 2 Mg & 3.5 Mg
Specification
- Product Name: Myezom
- Contains: Bortezomib
- Form: Vial
- Strength:2 Mg & 3.5 Mg
- Manufactured by :Dr.Reddys
NOTE:- Myezom is a prescription drug and should be used under proper medical guidance and advice. Do not share the medicine with others, since they may be suffering from a problem that is not effectively treated by this drug...............
Targeted Therapy for Blood Cancers
MYEZOM BORTEZOMIB 2 MG INJECTION is specifically formulated to target and interfere with cancer cell growth in patients diagnosed with Multiple Myeloma and Mantle Cell Lymphoma. By inhibiting proteasome activity, it disrupts the cellular processes vital for cancer cell survival and proliferation, delivering a focused approach to anticancer therapy.
Safe Handling and Administration
This medication is cytotoxic and requires careful handling by medical professionals. MYEZOM is supplied as a single-use vial containing lyophilized powder that must be reconstituted with an appropriate solvent prior to administration. Both intravenous and subcutaneous routes are used, as directed by a physician, ensuring safe and effective dosage delivery.
Storage and Shelf Life
MYEZOM should be stored at temperatures below 25C and protected from direct sunlight to maintain its effectiveness. The product has a shelf life of 24 months from the date of manufacture, provided it is properly sealed and stored according to the manufacturers guidelines. Always check expiry before use.
FAQs of MYEZOM BORTEZOMIB 2 MG INJECTION:
Q: How should MYEZOM BORTEZOMIB 2 MG INJECTION be administered?
A: MYEZOM should be reconstituted with an appropriate solvent by a healthcare professional and administered either intravenously or subcutaneously as prescribed by your doctor.Q: What is the main use of MYEZOM BORTEZOMIB INJECTION?
A: It is used for the treatment of adults with Multiple Myeloma and Mantle Cell Lymphoma, acting as an anticancer agent in these hematological conditions.Q: When should the injection be given?
A: The timing and frequency of administration are determined by your treating physician based on your medical condition and treatment schedule. Always follow your doctors instructions closely.Q: Where should MYEZOM INJECTION be stored?
A: Store the vial below 25C in a dry place, protected from light. Keep MYEZOM out of reach of children and only use if the vial is intact and within the expiry date.Q: What is the process for preparing MYEZOM before use?
A: Before administration, MYEZOM must be reconstituted with an appropriate sterile solvent by a medical professional, ensuring it is ready for injection as per prescribed guidelines.Q: What are the benefits of MYEZOM BORTEZOMIB INJECTION?
A: The primary benefit is its effectiveness in inhibiting cancer cell growth in conditions like Multiple Myeloma and Mantle Cell Lymphoma, offering a targeted treatment option that can improve patient outcomes when used correctly.Q: Is a prescription required to obtain MYEZOM BORTEZOMIB INJECTION?
A: Yes, MYEZOM is a prescription-only medicine and should be used under strict medical supervision due to its cytotoxic nature and specific dosing requirements.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Drugs & Medications Category
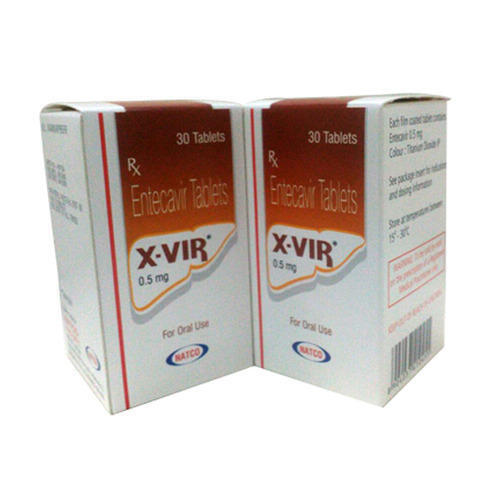
XVIR ENTECAVIR TABLETS
Minimum Order Quantity : 1 Bottle
Physical Form : Tablets
Dosage Guidelines : As prescribed by the healthcare professional
Storage Instructions : Store below 30C, protect from moisture and light
Dosage : 25mg
Origin of Medicine : India
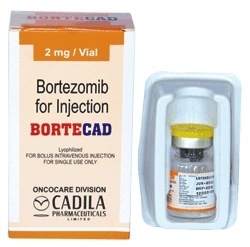
BORTECAD BORTEZOMIB INJECTION
Minimum Order Quantity : 1 Bottle
Physical Form : Liquid
Dosage Guidelines : Follow healthcare providers instructions
Storage Instructions : Store below 25C. Protect from light.
Dosage : As prescribed by physician
Origin of Medicine : Imported
HYDREA HYDROXYUREA CAPSULES
Physical Form : Other, Capsule
Dosage Guidelines : Take with or without food, whole capsule with water
Storage Instructions : Store below 25C, protect from moisture
Dosage : As directed by healthcare professional
Origin of Medicine : Allopathic
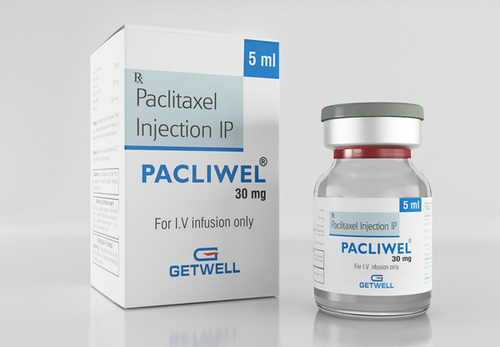
PACLIWEL PACLITAXEL INJECTION
Physical Form : Other, Liquid Injection
Dosage Guidelines : Intravenous infusion, follow physician guidance
Storage Instructions : Store below 25C, protect from light
Dosage : As prescribed by the physician
Origin of Medicine : Allopathic
 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry