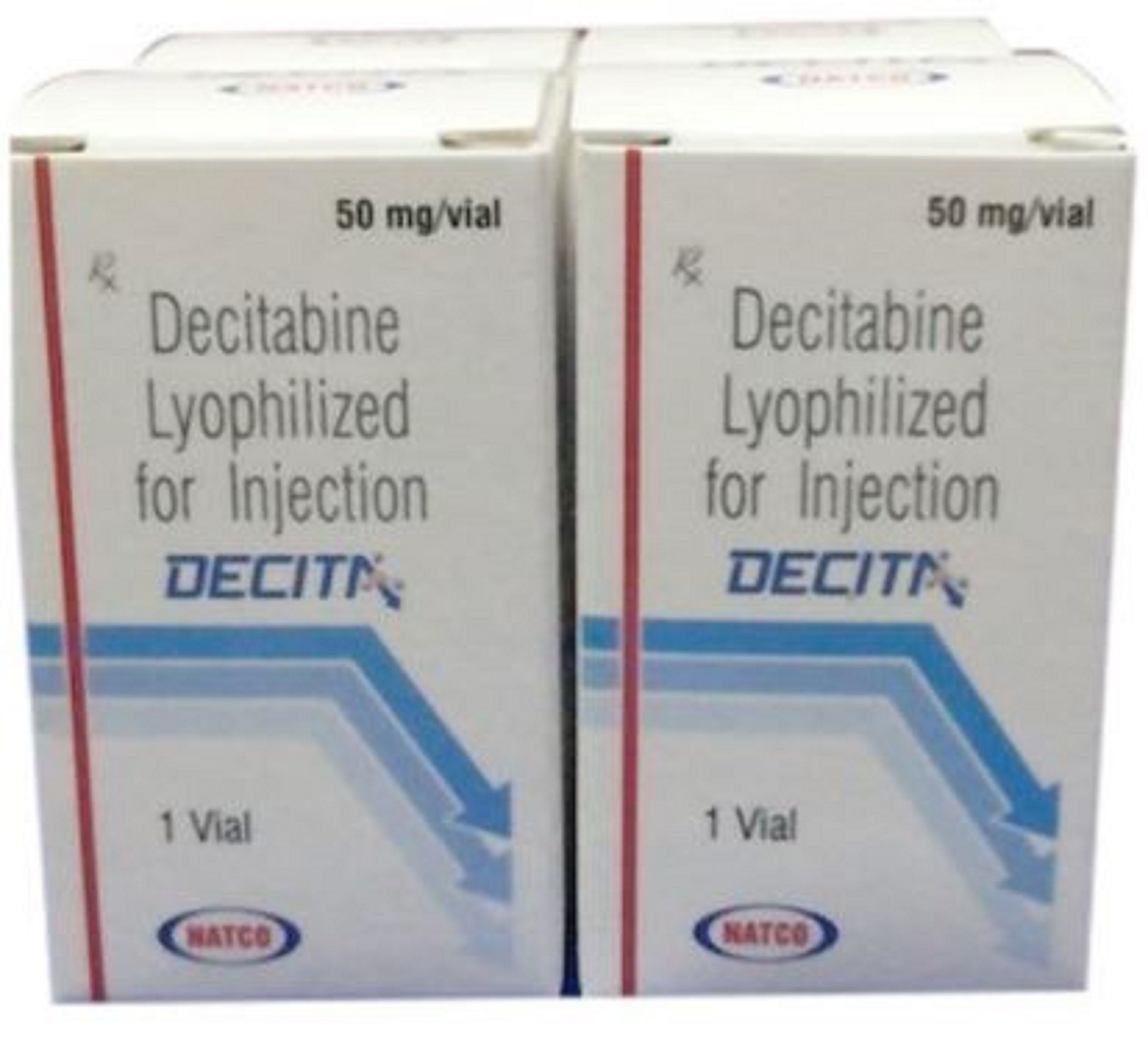
DECITA DECITABINE INJECTION
DECITA DECITABINE INJECTION Specification
- Indication
- Myelodysplastic Syndromes, Chronic Myelomonocytic Leukemia
- Dosage Form
- Injection
- Origin of Medicine
- Allopathic
- Pacakaging (Quantity Per Box)
- 1 vial per box
- Salt Composition
- Decitabine 50 mg
- Drug Type
- Injection
- Ingredients
- Decitabine
- Physical Form
- Liquid
- Function
- Antineoplastic
- Recommended For
- Anticancer therapy / Myelodysplastic Syndromes (MDS)
- Dosage
- As directed by physician
- Dosage Guidelines
- Intravenous infusion as prescribed
- Suitable For
- Adults
- Quantity
- 50 mg
- Storage Instructions
- Store below 30C, protect from light
- Appearance
- Clear, colorless to pale yellow solution
- Batch No.
- Provided on packaging
- Route of Administration
- Intravenous
- MRP
- As per local regulations
- Shelf Life
- 24 months
- Contraindications
- Pregnancy, lactation, hypersensitivity to decitabine
- Usage
- For hospital/institutional use only
- Warning
- To be administered under medical supervision only
- Marketed By
- Samarth Life Sciences Pvt Ltd
- Packaging Type
- Glass vial
- Therapeutic Class
- Anticancer / Cytotoxic agent
- Prescription Required
- Yes
DECITA DECITABINE INJECTION Trade Information
- Minimum Order Quantity
- 1 Bottle
- Packaging Details
- PACK OF 1 VIAL
- Main Domestic Market
- All India
About DECITA DECITABINE INJECTION
PRODUCT DETAILS
BRAND NAME: DECITA
CONTAINS: DECITABINE
STRENGTH: 50MG
PACKAGING SIZE: PACK OF 1 VIAL
MANUFACTURING BY: NATCO
Note :- DECITAIs APrescription Drug And Should Be Used Under Proper Medical Guidance And Advice.Do Not Share The Medicine With Others, Since They May Be Suffering FromAProblem That Is Not Effectively Treated By This Drug...................
Targeted Anticancer Therapy
DECITA Decitabine Injection delivers effective antineoplastic action against Myelodysplastic Syndromes and Chronic Myelomonocytic Leukemia. Its mechanism inhibits abnormal cell growth and helps restore normal blood cell production, supporting better health outcomes in eligible adults. Treatment is strictly as per a physicians direction, ensuring optimal benefit and safety.
Safe Handling and Administration
This injection is intended strictly for intravenous use in hospitals or medical institutions, supervised by healthcare professionals. Each vial is single-use, minimizing contamination risks. Proper storagebelow 30C and away from lightpreserves its efficacy for up to 24 months.
FAQs of DECITA DECITABINE INJECTION:
Q: How should DECITA Decitabine Injection be administered?
A: DECITA Decitabine Injection must be administered intravenously under the close supervision of qualified medical personnel in a hospital or institutional setting, as prescribed by your physician.Q: What conditions does this medicine treat?
A: DECITA is recommended for adults in the management of Myelodysplastic Syndromes (MDS) and Chronic Myelomonocytic Leukemia, functioning as an antineoplastic agent to control abnormal cell proliferation.Q: When is it contraindicated to use DECITA Decitabine Injection?
A: The injection is contraindicated in individuals who are pregnant, breastfeeding, or have hypersensitivity to Decitabine. Always inform your doctor about any relevant conditions before starting treatment.Q: Where should the injection be stored and for how long?
A: The vial should be stored below 30C in a place protected from light. DECITA has a shelf life of 24 months from the date of manufacture, as indicated on the packaging.Q: What are the main benefits of DECITA Decitabine Injection?
A: DECITA provides targeted and effective anticancer action in adults with MDS and related leukemia, helping to manage abnormal blood cell growth when treatment is administered as prescribed.Q: How is the dosage of DECITA determined?
A: Dosage and treatment regimen for DECITA are determined exclusively by a physician based on individual patient profiles and clinical indications.


Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Drugs & Medications Category
TENVIR TENOFOVIR TABLETS
Minimum Order Quantity : 1 Bottle
Suitable For : Adults
Quantity : 30 tablets per bottle
Drug Type : Other, Allopathic
Recommended For : Adults and adolescents above 12 years
Function : Other, Antiviral medication for the treatment of HIV infection
Mesalzer 1.2 gm Tablets
Minimum Order Quantity : 1 Bottle
Suitable For : Adults
Quantity : 10 x 10 Tablets
Drug Type : Other, Prescription
Recommended For : For treatment of mild to moderate ulcerative colitis
Function : Other, Antiinflammatory; used in inflammatory bowel disease
TRIODAY LAMIVUDINE TENOVOFIR TABLETS
Minimum Order Quantity : 1 Bottle
Suitable For : Adults
Quantity : 30 Tablets per box
Drug Type : Other, Allopathic
Recommended For : HIV Infection
Function : Other, Antiretroviral therapy
GEFTIB GEFTINIB TABLETS
Suitable For : Adults
Quantity : 30 Tablets
Drug Type : Other Types
Recommended For : Nonsmall cell lung cancer
Function : Other, Anticancer, EGFR inhibitor
 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry