Alimta
Alimta Specification
- Dosage Form
- Lyophilized Powder for Injection
- Origin of Medicine
- Imported
- Salt Composition
- Pemetrexed Disodium
- Pacakaging (Quantity Per Box)
- 1 vial per box
- Indication
- Malignant pleural mesothelioma and non-small cell lung cancer
- Drug Type
- Allopathic
- Ingredients
- Pemetrexed Disodium Hemipenta Hydrate
- Physical Form
- Injection
- Function
- Antineoplastic (anti-cancer)
- Recommended For
- Anti-cancer / Used in the treatment of malignant pleural mesothelioma and non-small cell lung cancer
- Dosage
- 500 mg/vial
- Dosage Guidelines
- As directed by the oncologist
- Suitable For
- Adults
- Quantity
- 1 vial
- Storage Instructions
- Store below 25C, protect from light
- Brand Name
- Alimta
- Marketed Strength
- 500 mg
- Prescription
- Prescription only medicine
- Warning
- For single use only; discard unused portion
- Registration
- Registered under pharmaceutical drug regulations
- Reconstitution
- To be reconstituted with 20 ml 0.9% Sodium Chloride Injection
- Administration Route
- Intravenous (IV) use after reconstitution
Alimta Trade Information
- Minimum Order Quantity
- 1 Bottle
- Main Domestic Market
- All India
About Alimta
Brand Name : Alimta
Content : Pemetrexed For Injection
Strength: Pemetrexed For Injection 500mg
Packing : 500mg
Type : Injection
Manufactured by: Alkem Laboratories Lt............
Powerful Cancer Treatment
Alimta delivers targeted chemotherapy for patients with malignant pleural mesothelioma and non-small cell lung cancer. Its advanced formulation works by interfering with cancer cell growth, helping to manage these aggressive conditions. Regular administration under oncologist guidance ensures optimal efficacy and safety.
Professional Preparation and Administration
Alimta is supplied as a 500 mg vial lyophilized powder and requires precise reconstitution with 20 ml of 0.9% Sodium Chloride Injection. Administration is via intravenous infusion in a clinical setting, ensuring accurate dosing and patient monitoring during treatment sessions.
Stringent Safety and Storage Measures
To maintain effectiveness and safety, Alimta must be stored below 25C and protected from light. As a single-use medication, any unused portion should be safely discarded. Adherence to these guidelines preserves medicine quality and reduces risk of contamination.
FAQs of Alimta:
Q: How is Alimta prepared and administered to patients?
A: Alimta is supplied as a lyophilized powder for injection. Before use, it is reconstituted with 20 ml of 0.9% Sodium Chloride Injection and then administered intravenously by trained healthcare professionals.Q: What conditions is Alimta prescribed for?
A: Alimta is recommended for adults in the treatment of malignant pleural mesothelioma and non-small cell lung cancer, functioning as a chemotherapeutic agent to slow cancer progression.Q: When should Alimta be used during cancer treatment?
A: Alimta is used as directed by an oncologist, typically as part of a prescribed chemotherapy regimen for eligible adults diagnosed with the indicated cancers.Q: Where should Alimta be stored before use?
A: Unopened vials of Alimta must be stored below 25C and protected from light to ensure the medications integrity and effectiveness.Q: What is the process for using Alimta in clinical practice?
A: After reconstitution, Alimta is administered via intravenous infusion. The remaining unused portion must be discarded as it is a single-use formulation, and all procedures are handled by medical professionals.Q: What benefits does Alimta offer to patients?
A: Alimta provides antineoplastic action, efficiently targeting cancer cells to help manage mesothelioma and non-small cell lung cancer, improving treatment outcomes under medical supervision.Q: Is Alimta suitable for self-administration?
A: No, Alimta requires preparation and intravenous administration by trained healthcare personnel in a medical facility. Self-administration is not recommended or permitted.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Drugs & Medications Category
TEMOREL TEMOZOLOMIDE CAPSULES
Dosage : As directed by physician
Physical Form : Other, Capsule
Dosage Guidelines : Take on an empty stomach, whole without breaking or chewing
Origin of Medicine : Allopathic
Ingredients : Temozolomide
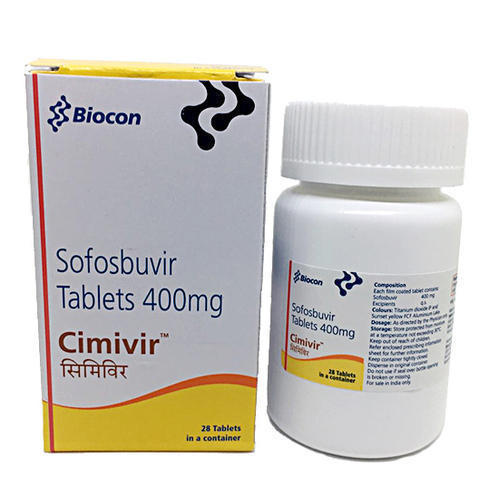
CIMIVIR SOFOSBUVIR TABLETS
Dosage : 400 mg
Physical Form : Tablets
Dosage Guidelines : As directed by physician
Origin of Medicine : India
Ingredients : Sofosbuvir
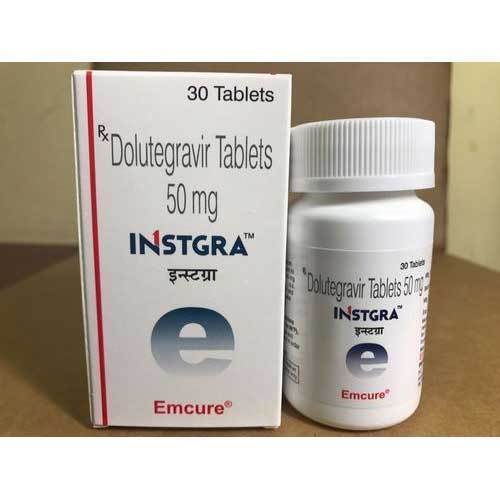
INSTGRA DOLUTEGRAVIR TABLETS
Dosage : 50 mg
Physical Form : Tablets
Dosage Guidelines : As directed by the physician
Origin of Medicine : India
Ingredients : Dolutegravir
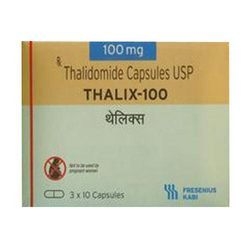
THALIX-THALIDOMIDE CAPSULES
Dosage : As prescribed by physician
Physical Form : Capsules
Dosage Guidelines : Follow physicians instruction
Origin of Medicine : India
Ingredients : Thalidomide
 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry